|
6/1/2023 0 Comments Largest atomic radius
Know about Electron Gain Enthalpy? 3) Metallic RadiusĪ metal lattice or crystal consists of positive kernels or metal ions arranged in a definite pattern in a sea of mobile valence electrons. Therefore, the Van der Waals radius of the chlorine atom is 180 pm. The magnitude of the Van der Waals radius is dependent on the packing of the atoms when the element is in the solid-state.įor example, the internuclear distance between two adjacent chlorine atoms of the two neighboring molecules in the solid-state is 360 pm. It is one half the distance between the nuclei of two identical non-bonded isolated atoms or two adjacent identical atoms belonging to two neighboring molecules of an element in the solid-state. The internuclear distance between two bonded atoms is called the bond length. Therefore, r covalent = ½ (internuclear distance between two bonded atoms). 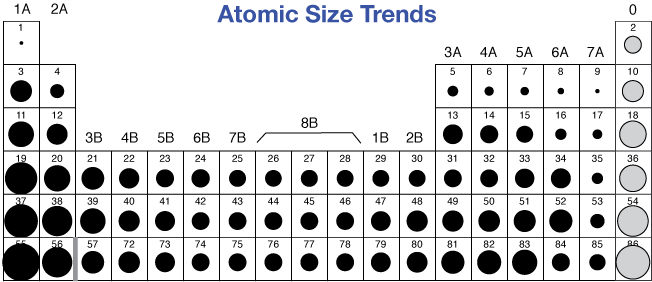
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
